What do your Mitochondria influence Quality of Life?
Let’s start with some key definitions to understand the high and why…
Mitochondria are double-membraned organelles found in most eukaryotic cells.
They are often described as the “powerhouses of the cell” because they generate the majority of the cell’s supply of adenosine triphosphate (ATP), which is used as a source of chemical energy.
In addition to energy production, mitochondria are involved in various other processes, including signaling, cellular differentiation, cell death, as well as the control of the cell cycle and cell growth.
Definition of Mitochondrial Biogenesis
Mitochondrial biogenesis is the process by which cells increase their individual mitochondrial mass and copy number to increase energy production. This process involves the coordinated expression of nuclear and mitochondrial genomes and the synthesis of mitochondrial proteins, lipids, and other essential components. Key regulators of mitochondrial biogenesis include peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), nuclear respiratory factors (NRFs), and mitochondrial transcription factor A (TFAM).
Definition of Mitochondrial Capacity and How to Increase It
Mitochondrial capacity refers to the ability of mitochondria to produce ATP through oxidative phosphorylation. This capacity is influenced by the number, size, and efficiency of mitochondria within a cell.
To increase mitochondrial capacity, one can:
- Exercise: Regular physical activity, especially aerobic exercises like running, cycling, and swimming, stimulates mitochondrial biogenesis and enhances the efficiency of existing mitochondria.
- Diet: Nutritional strategies, such as calorie restriction and a diet rich in polyphenols (e.g., from berries, nuts, and dark chocolate), can promote mitochondrial biogenesis.
- Supplements: Certain supplements, such as coenzyme Q10, omega-3 fatty acids, and resveratrol, may support mitochondrial function.
- Cold Exposure: Regular exposure to cold temperatures can stimulate the production of brown adipose tissue, which is rich in mitochondria.
Definition of Mitochondrial Density and How to Improve It

- Mitochondrial density refers to the number of mitochondria per unit volume of the cell.
- Higher mitochondrial density generally correlates with increased cellular energy capacity and improved metabolic efficiency.
To improve mitochondrial density:
- Exercise: Similar to increasing mitochondrial capacity, consistent aerobic exercise can enhance mitochondrial density.
- Intermittent Fasting: Periods of fasting can trigger adaptive cellular responses that increase mitochondrial density.
- Nutrient Timing: Ensuring adequate nutrient intake around periods of high energy demand (e.g., before and after exercise) can support mitochondrial biogenesis and density.
Definition of Mitophagy and How to Improve It
Mitophagy is the selective degradation of mitochondria by autophagy. It is a quality control mechanism that removes damaged or dysfunctional mitochondria, thereby maintaining cellular health. This process involves the encapsulation of defective mitochondria by autophagosomes, which then fuse with lysosomes to degrade the mitochondrial contents.
 To improve mitophagy:
To improve mitophagy:
- Exercise: Physical activity has been shown to enhance mitophagy, helping maintain mitochondrial quality.
- Diet: Certain diets, like calorie restriction or ketogenic diets, can stimulate mitophagy.
- Phytochemicals: Compounds such as polyphenols (e.g., resveratrol, quercetin) can activate pathways that promote mitophagy.
- Supplements: Some supplements, such as spermidine, have been shown to enhance autophagy and mitophagy.
In summary, mitochondria are crucial organelles for cellular energy production, and their function and efficiency can be enhanced through various lifestyle interventions, including exercise, diet, and certain supplements.
Exercise and Mitochondrial Biogenesis
To create new mitochondria, specific types, intensities, durations, and frequencies of exercise are particularly effective. Here are the details:
Type of Exercise
- Aerobic Exercise: Activities such as running, cycling, swimming, and brisk walking are most effective at stimulating mitochondrial biogenesis. These exercises rely on sustained, rhythmic movements that increase oxygen consumption and demand energy production over extended periods.
Intensity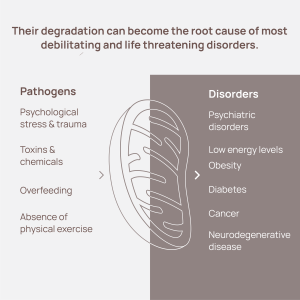
- Moderate to High Intensity: Exercises performed at 60-85% of your maximum heart rate are ideal. High-intensity interval training (HIIT), which alternates short bursts of intense exercise with periods of rest or low activity, is particularly potent for mitochondrial biogenesis.
Duration
- At least 30 minutes per session: Sustained aerobic activities for at least 30 minutes per session are necessary to stimulate the cellular pathways that promote mitochondrial biogenesis.
Frequency
- 3-5 times per week: Regular exercise sessions, spaced throughout the week, ensure continuous stimulation of mitochondrial biogenesis. Consistency is key for long-term adaptations.
Exercise for Improving Mitochondrial Capacity
Type of Exercise
- Aerobic and HIIT: Both continuous aerobic exercise and HIIT are effective at improving mitochondrial capacity.
Intensity
- Moderate to High Intensity: Exercise performed at moderate to high intensity challenges the mitochondria, improving their efficiency and capacity.
Duration and Frequency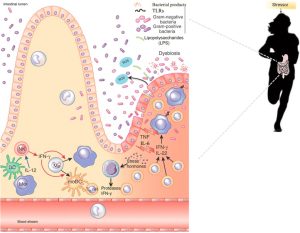
- Similar to those for biogenesis: Regular, consistent exercise sessions of at least 30 minutes, 3-5 times per week.
Definitions
Mitochondrial Capacity
Mitochondrial capacity refers to the ability of mitochondria to produce ATP through oxidative phosphorylation. It is a measure of how effectively mitochondria can meet the energy demands of the cell. This capacity is influenced by the number, size, and efficiency of mitochondria.
Mitochondrial Function
Mitochondrial function encompasses the overall performance of mitochondria, including ATP production, regulation of cellular metabolism, generation of reactive oxygen species (ROS), and involvement in cellular signaling and apoptosis. Healthy mitochondrial function is crucial for energy metabolism and cellular health.
Mitochondrial Density
Mitochondrial density refers to the number of mitochondria per unit volume of the cell. Higher mitochondrial density generally correlates with increased cellular energy capacity and improved metabolic efficiency. Increasing mitochondrial density typically involves stimulating mitochondrial biogenesis through regular exercise and other interventions.
Summary
- To create new mitochondria and improve their capacity, engaging in regular aerobic exercise and high-intensity interval training (HIIT) is essential.
- Moderate to high-intensity workouts performed for at least 30 minutes, 3-5 times per week, stimulate the necessary adaptations.
- Understanding mitochondrial capacity, function, and density helps to appreciate the cellular mechanisms underlying these benefits and guides effective exercise regimens for enhancing cellular health.
What is The GOLDILOCKS Effect for the ideal dose of exercise?
Research on the Ideal Dose of HIIT and Zone Two Exercise
To stimulate mitochondrial biogenesis and improve mitochondrial capacity without overtraining or causing excessive oxidative stress, both High-Intensity Interval Training (HIIT) and Zone Two exercise (moderate-intensity aerobic exercise) are highly effective.
Here’s what research suggests about the ideal dose for each:
High-Intensity Interval Training (HIIT)
HIIT involves short bursts of intense exercise followed by recovery periods.
- Frequency:
- 2-3 times per week.
- Duration:
- Each session should last about 20-30 minutes.
- Intensity:
- Work intervals should be performed at 85-95% of maximum heart rate.
- Recovery intervals can be at 50-70% of maximum heart rate.
- Zone 4/1 for HIIT
- Zone 5/1 for SIT
- Work-to-Rest Ratio:
- Common protocols use a work-to-rest ratio of 1:1 or 2:1. For example, 30 seconds of intense exercise followed by 30 seconds of rest, or 30 seconds of intense exercise followed by 15 seconds of rest.
- Total Number of Intervals:
- Typically 6-10 intervals per session.
Research Findings:
- Studies indicate that as few as three HIIT sessions per week can significantly enhance mitochondrial biogenesis and capacity.
- A study by Gibala et al. found that 6 sessions of HIIT over 2 weeks increased muscle oxidative capacity and doubled the endurance capacity of participants.
- Another study by Little et al. reported that HIIT protocols with shorter duration (e.g., 10-minute sessions including warm-up and cool-down) are effective in stimulating mitochondrial adaptations.
Zone Two Exercise
Zone Two exercise involves moderate-intensity, steady-state aerobic exercise.
- Frequency:
- 3-5 times per week.
- Duration:
- Each session should last 45-60 minutes.
- Intensity:
- Exercise should be performed at 60-70% of maximum heart rate.
- This intensity corresponds to a level where you can maintain a conversation comfortably (known as the “talk test”).
- Steady-State:
- The exercise should be continuous and maintain a steady heart rate within Zone Two.
Research Findings:
- Zone Two exercise is effective for enhancing mitochondrial density and function with lower oxidative stress compared to higher-intensity workouts.
- A study by Holloszy and Coyle demonstrated that prolonged moderate-intensity exercise leads to significant increases in mitochondrial content and respiratory capacity in muscle.
- Additional research suggests that consistent Zone Two training improves oxidative phosphorylation and reduces lactate production, which is beneficial for endurance and metabolic health.
Balancing HIIT and Zone Two Exercise
Combination Training:
- Weekly Schedule: A balanced training schedule might include 2 days of HIIT and 3 days of Zone Two exercise.
- Recovery: Ensure adequate rest and recovery between high-intensity sessions to prevent overtraining and manage oxidative stress.
- Monitoring: Pay attention to signs of overtraining, such as prolonged fatigue, decreased performance, and increased resting heart rate. Adjust the training intensity and volume accordingly.
Managing Oxidative Stress
- Antioxidant-Rich Diet: Consume a diet rich in antioxidants to help mitigate exercise-induced oxidative stress.
- Adequate Sleep: Ensure sufficient sleep for recovery and mitochondrial repair.
- Hydration: Stay well-hydrated to support metabolic processes and recovery.
Summary
- Both HIIT and Zone Two exercises are effective for stimulating mitochondrial biogenesis and improving mitochondrial capacity.
- By combining these exercise modalities and adhering to recommended doses, individuals can optimize mitochondrial adaptations while minimizing the risk of overtraining and oxidative stress.

Exercise Effectiveness for Mitochondrial Biogenesis and Capacity
Mitochondrial Biogenesis
High-Intensity Interval Training (HIIT) is generally considered more effective for mitochondrial biogenesis compared to moderate-intensity continuous training (like Zone Two exercise). Here’s why:
- Intensity: HIIT involves short bursts of high-intensity exercise that significantly stress the muscles and energy systems, triggering robust adaptive responses.
- AMPK Activation: High-intensity efforts rapidly deplete ATP, activating AMP-activated protein kinase (AMPK), a key regulator of mitochondrial biogenesis.
- PGC-1α Activation: HIIT strongly induces the expression of peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), a critical transcriptional coactivator involved in the formation of new mitochondria.
- Research Evidence: Studies have shown that even short-term HIIT can significantly increase markers of mitochondrial biogenesis. For example, Gibala et al. demonstrated that 6 sessions of HIIT over two weeks substantially increased muscle oxidative capacity.
Key Protocols:
- Work-to-rest ratios of 1:1 or 2:1 (e.g., 30 seconds high-intensity followed by 30 seconds rest).
- Sessions lasting 20-30 minutes.
- Frequency of 2-3 times per week.
Mitochondrial Capacity
Moderate-Intensity Continuous Training (Zone Two Exercise) is particularly effective at improving mitochondrial capacity.
Here’s why:
- Sustained Effort: Prolonged, steady-state exercise enhances the efficiency of the mitochondria and their ability to sustain ATP production over extended periods.
- Oxidative Phosphorylation: Zone Two training primarily utilizes aerobic pathways, enhancing the capacity of mitochondria to produce energy through oxidative phosphorylation.
- Low Oxidative Stress: Moderate intensity exercises induce less oxidative stress compared to high-intensity efforts, allowing for consistent improvements in mitochondrial function without excessive damage.
- Research Evidence: Studies, such as those by Holloszy and Coyle, have shown significant improvements in mitochondrial content and respiratory capacity with consistent moderate-intensity training.
Key Protocols: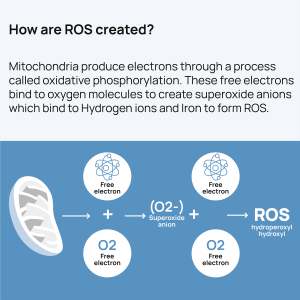
- Steady-state exercise at 60-70% of maximum heart rate.
- Sessions lasting 45-60 minutes.
- Frequency of 3-5 times per week.
Conclusion
- For Mitochondrial Biogenesis: HIIT is more effective due to its ability to strongly activate pathways that promote the creation of new mitochondria.
- For Mitochondrial Capacity: Zone Two exercise is particularly beneficial, enhancing the efficiency and capacity of existing mitochondria through sustained aerobic activity.
Balancing Both
For optimal results, incorporating both HIIT and Zone Two exercise into a training regimen can provide comprehensive benefits:
- HIIT: 2-3 times per week to stimulate mitochondrial biogenesis.
- Zone Two: 3-5 times per week to improve mitochondrial capacity and overall endurance.
This balanced approach ensures robust mitochondrial health, enhancing both the number and efficiency of mitochondria.
 How PNOE Metabolism Testing Can Guide Training for Improved Mitochondrial Function
How PNOE Metabolism Testing Can Guide Training for Improved Mitochondrial Function
PNOE Metabolism Testing is a comprehensive metabolic analysis that evaluates various aspects of an individual’s metabolic function. This testing provides detailed insights into how efficiently the body uses oxygen and produces energy during exercise, which is crucial for optimizing training programs to enhance mitochondrial function.
Here’s how PNOE Metabolism Testing can help determine the appropriate type, frequency, and intensity of training:
Assessing Aerobic Capacity and Efficiency
Aerobic capacity is a key indicator of mitochondrial function. PNOE testing measure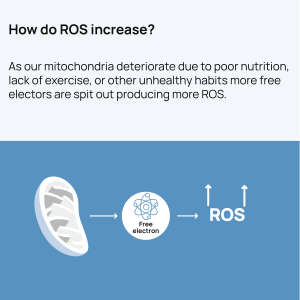 s:
s:
- VO2 Max: The maximum amount of oxygen the body can utilize during intense exercise. A higher VO2 max indicates better mitochondrial capacity.
- VO2 at different intensities: How oxygen consumption changes at various exercise intensities.
Guidance:
- For enhancing mitochondrial capacity: Training in zones that improve aerobic efficiency (Zone Two) will be identified.
- For mitochondrial biogenesis: High-intensity intervals that challenge the upper limits of oxygen consumption will be recommended.
Identifying Anaerobic Threshold
Anaerobic threshold (AT), or where the fuel source changes from fat fuel, is the point at which lactate begins to accumulate in the blood. This threshold is a marker of the balance between aerobic and anaerobic energy production, directly linked to mitochondrial efficiency.
Guidance:
- Training Zones: Knowing the AT helps determine the exact intensities at which to train to stay within aerobic zones (improving mitochondrial density and function) versus anaerobic zones (stimulating mitochondrial biogenesis).
- Customization: Training intensities can be precisely tailored to work just below, at, or above the AT depending on the desired mitochondrial adaptation.
Measuring Respiratory Exchange Ratio (RER)
RER provides insights into the substrates (carbohydrates vs. fats) being used for energy at different intensities.
- An RER close to 1 indicates predominant carbohydrate usage (anaerobic metabolism).
- An RER closer to 0.7 indicates predominant fat usage (aerobic metabolism).
Guidance:
- Optimizing Fuel Utilization: Training recommendations can be made to shift the RER towards more efficient fat utilization, enhancing mitochondrial function and endurance.
Evaluating Recovery and Fatigue
Recovery metrics are crucial for avoiding overtraining, which can impair mitochondrial function. PNOE testing assesses:
- Heart rate recovery: How quickly the heart rate returns to baseline after exercise.
- Ventilatory efficiency: The effectiveness of the respiratory system during recovery.
Guidance:
- Balancing Intensity: Insights into recovery allow for better planning of high-intensity vs. moderate-intensity sessions to ensure adequate recovery and avoid excessive oxidative stress.
- Frequency Adjustments: Determining the optimal frequency of HIIT and Zone Two exercises based on individual recovery rates.
Tracking Progress
PNOE testing can be repeated periodically to track changes in metabolic efficiency and mitochondrial function over time.
Guidance:
- Adjusting Programs: As mitochondrial function improves, training programs can be adjusted to maintain progression and avoid plateaus.
- Personalized Adaptations: Continuous monitoring ensures that training remains aligned with current metabolic capabilities and goals.
Practical Implementation
Using PNOE Data for Training Plans:
- Initial Assessment: Conduct a baseline PNOE test to determine current aerobic capacity, anaerobic threshold, RER, and recovery metrics.
- Program Design:
- HIIT Sessions: Based on VO2 max and AT data, design HIIT sessions at 85-95% of maximum heart rate, focusing on work-to-rest ratios that match individual metabolic responses.
- Zone Two Training: Implement moderate-intensity sessions (60-70% of max heart rate) to improve aerobic efficiency and mitochondrial density.
- Monitor and Adjust:
- Regularly retest to monitor progress and adjust training intensities, durations, and frequencies.
- Use recovery metrics to ensure adequate rest and avoid overtraining.
By leveraging the detailed metabolic insights provided by PNOE testing, athletes and trainers can create highly personalized and effective training programs that optimize mitochondrial function, enhancing overall performance and health.
What is the CELL DANGER RESPONSE Theory… and relationship to mitochondria function?
The Cell Danger Response (CDR) theory is a concept in biology that suggests cells react to danger or stress through a coordinated metabolic response designed to protect the organism. This theory, proposed by Dr. Robert Naviaux, posits that cells under threat, whether from infections, toxins, physical injury, or other stressors, undergo a series of metabolic and molecular changes aimed at ensuring survival and restoring homeostasis.
Key Points of the Cell Danger Response Theory
- Metabolic Shift: When a cell detects danger, it shifts its metabolism from a state focused on growth and normal function to one that prioritizes defense and repair. This often involves a switch from oxidative phosphorylation (aerobic energy production in mitochondria) to glycolysis (anaerobic energy production).
- Communication: Cells undergoing the CDR release signaling molecules like ATP, purines, and reactive oxygen species (ROS) to alert neighboring cells and the immune system about the presence of a threat. These molecules act as “danger signals.“
- Protective Mechanisms: The CDR involves the activation of various protective mechanisms, including autophagy (the process of cleaning out damaged cellular components), increased production of antioxidants, and the expression of stress proteins.
- Temporary State: The CDR is meant to be a temporary state that resolves once the threat is eliminated. Cells then shift back to their normal metabolic functions, restoring homeostasis.
- Chronic Activation: Problems arise when the CDR is chronically activated due to persistent stressors or an inability to resolve the initial threat. Chronic CDR can lead to sustained inflammation, cellular dysfunction, and contribute to the development of chronic diseases and conditions such as autism, chronic fatigue syndrome, and various neurodegenerative diseases.
- Role of Mitochondria: Mitochondria play a central role in the CDR because they are both the powerhouses of the cell and key sensors of cellular stress. Dysfunctional mitochondria can perpetuate the danger response, leading to prolonged or maladaptive CDR.
Implications of the Cell Danger Response Theory
- Disease Understanding: The CDR theory provides a framework for understanding how various chronic diseases may develop from an initial acute stressor or injury that fails to resolve properly.
- Treatment Approaches: Therapeutic strategies might focus on identifying and removing the persistent stressors, supporting mitochondrial function, and promoting the resolution of the CDR to restore normal cellular function.
- Research and Medicine: The theory encourages a holistic view of disease, considering metabolic, environmental, and genetic factors, and highlights the importance of mitochondrial health in disease prevention and treatment.
Conclusion
The Cell Danger Response theory is a compelling model that explains how cells react to threats and how chronic diseases may arise from prolonged or unresolved cellular stress responses. By focusing on the role of mitochondria and the importance of restoring cellular homeostasis, the CDR theory provides valuable insights for developing new treatments and preventive measures for various chronic conditions.
Too much of anything… Chronic Stressors
Impact of Oxidative Stress from Excessive Exercise on Mitochondrial Health and Longevity, Chronic Fatigue, and Chronic Inflammation
Oxidative stress occurs when there is an imbalance between the production of reactive oxygen species (ROS) and the body’s ability to detoxify these reactive intermediates or repair the resulting damage.
While ROS are a natural byproduct of aerobic metabolism and play a role in cell signaling, excessive levels can be detrimental.
Impact on Mitochondrial Health and Longevity
Mitochondrial Damage:
- Excess ROS Production: During excessive exercise, mitochondria produce large amounts of ROS.
- High levels of ROS can damage mitochondrial DNA, proteins, and lipids, impairing mitochondrial function.
- Mitochondrial Dysfunction: Accumulated damage can lead to mitochondrial dysfunction, where the efficiency of ATP production is reduced.
- This dysfunction can result in a decreased ability to meet the energy demands of cells.
- Mitochondrial Biogenesis Inhibition: Chronic oxidative stress can impair the signaling pathways involved in mitochondrial biogenesis, such as the PGC-1α pathway, reducing the cell’s ability to produce new mitochondria.
- Apoptosis: Severe mitochondrial damage can trigger apoptotic pathways, leading to cell death and potentially contributing to tissue degeneration.
Longevity:
- Accelerated Aging: Persistent oxidative damage to mitochondria is linked to accelerated aging processes.
- Dysfunctional mitochondria can contribute to cellular senescence and reduced cellular repair mechanisms, leading to a decline in tissue and organ function over time.
Impact on Chronic Fatigue
Energy Production:
- Reduced ATP Availability: Mitochondrial dysfunction leads to a decrease in ATP production, the primary energy currency of the cell.
- This energy deficit is particularly impactful in muscle cells, contributing to feelings of fatigue and reduced endurance.
- Impaired Muscle Function: Damaged mitochondria can impair muscle function and recovery, leading to muscle weakness and persistent fatigue even after rest.
Systemic Effects:
- Chronic Fatigue Syndrome (CFS): There is evidence linking mitochondrial dysfunction and oxidative stress to chronic fatigue syndrome. Individuals with CFS often show signs of impaired mitochondrial function and elevated oxidative stress markers.
Impact on Chronic Inflammation
Inflammatory Pathways:
- NF-κB Activation: Excessive ROS can activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), a key regulator of inflammatory responses. This activation leads to the production of pro-inflammatory cytokines.
- Inflammasome Activation: Oxidative stress can also activate inflammasomes, multi-protein complexes that trigger inflammatory processes, further contributing to chronic inflammation.
Cellular Damage:
- Immune Cell Dysfunction: Chronic oxidative stress can impair the function of immune cells, leading to an inappropriate or exaggerated immune response. This dysfunction can exacerbate inflammation and delay the resolution of inflammatory processes.
- Tissue Damage: Persistent inflammation can cause ongoing tissue damage and fibrosis, leading to further oxidative stress and creating a vicious cycle of inflammation and damage.
Mitigation Strategies
Balancing Exercise:
- Moderation: Engage in moderate-intensity exercise and avoid excessive high-intensity training without adequate recovery.
- Periodization: Implement periodization in training programs to ensure balanced workloads and sufficient rest periods.
Antioxidant Support:
- Diet: Consume a diet rich in antioxidants (e.g., fruits, vegetables, nuts) to help neutralize excess ROS.
- Supplements: Consider antioxidant supplements (e.g., vitamins C and E, coenzyme Q10) as recommended by healthcare providers.
Recovery Practices:
- Rest and Sleep: Ensure adequate sleep and rest between training sessions to allow for proper recovery and repair.
- Hydration and Nutrition: Maintain proper hydration and nutrition to support metabolic processes and reduce oxidative stress.
Monitoring and Adjustment:
- Biomarkers: Monitor biomarkers of oxidative stress and inflammation (e.g., C-reactive protein, malondialdehyde) to tailor training programs and interventions.
- Personalized Training: Use tools like PNOE Metabolism Testing to personalize training intensity, duration, and frequency based on individual responses and recovery rates.
Excessive exercise-induced oxidative stress can significantly impact mitochondrial health, contributing to mitochondrial dysfunction, reduced longevity, chronic fatigue, and chronic inflammation. Balancing exercise intensity and duration, supporting antioxidant defenses, and ensuring proper recovery are essential strategies to mitigate these negative effects and promote overall cellular and mitochondrial health
What are the most nutrient dense anti-oxidant rich foods from plants and animal sources?
Yes, there are several non-plant foods that are nutrient-dense and high in antioxidants. While plant-based foods are typically the richest sources of antioxidants, certain animal-derived and other non-plant foods also contain notable amounts of antioxidants and other beneficial nutrients. Here are some examples:
1. Fish and Seafood
- Salmon: Rich in omega-3 fatty acids, particularly EPA and DHA, which have antioxidant properties and anti-inflammatory effects.
- Shellfish (e.g., shrimp, crab): Provide selenium, zinc, and astaxanthin, a potent antioxidant.
- Oysters: High in zinc, selenium, and copper, which are essential for antioxidant enzyme function.
2. Eggs
- Yolk: Contains lutein and zeaxanthin, carotenoid antioxidants that support eye health.
- Omega-3 Enriched Eggs: May have higher levels of omega-3 fatty acids and vitamin E.
3. Meat
- Lean Beef: Contains zinc, selenium, and vitamin B12, which support antioxidant enzyme function.
- Turkey and Chicken: Provide selenium and B vitamins, including niacin and riboflavin.
4. Dairy Products
- Yogurt: Contains probiotics and some antioxidants like vitamin E and selenium.
- Cheese: Provides calcium, phosphorus, and vitamin A, which has antioxidant properties.
5. Organ Meats
- Liver: Particularly beef liver, is rich in vitamins A, B12, and folate, as well as minerals like iron and copper, which support antioxidant enzyme function.
6. Other Animal-Derived Products
- Bone Broth: Contains collagen, gelatin, and amino acids that support gut health and immune function.
- Honey: Contains flavonoids and phenolic acids with antioxidant properties.
7. Fermented Foods
- Kimchi and Sauerkraut: Fermented vegetables that provide probiotics and some antioxidant compounds.
8. Mushrooms
- While technically not plants, mushrooms are fungi and are rich in antioxidants like ergothioneine and selenium.
9. Bee Products
- Propolis: Contains flavonoids with antioxidant and anti-inflammatory properties.
- Royal Jelly: Rich in vitamins, minerals, and antioxidants.
10. Seaweed
- Rich in vitamins (e.g., vitamin C) and minerals (e.g., iodine) with some antioxidant properties.

Nutrient-dense foods rich in antioxidants play a crucial role in neutralizing free radicals and reducing oxidative stress, which can protect mitochondrial health and overall cellular function.
Here are some of the top foods known for their high antioxidant content -plant based.
1. Berries
- Blueberries: Rich in anthocyanins, vitamins C and K, and fiber.
- Strawberries: High in vitamin C, manganese, and a variety of antioxidants such as ellagic acid.
- Raspberries: Contain vitamin C, quercetin, and ellagic acid.
- Blackberries: Packed with vitamins C and K, fiber, and anthocyanins.
2. Nuts and Seeds
- Walnuts: High in omega-3 fatty acids, vitamin E, and polyphenols.
- Almonds: Rich in vitamin E, magnesium, and fiber.
- Chia Seeds: Contain omega-3 fatty acids, fiber, and various antioxidants.
- Flaxseeds: High in lignans, fiber, and omega-3 fatty acids.
3. Leafy Greens
- Kale: Contains vitamins A, C, and K, as well as quercetin and kaempferol.
- Spinach: Rich in vitamins A, C, and K, folate, iron, and lutein.
- Swiss Chard: Provides vitamins A, C, and K, magnesium, and antioxidants like beta-carotene.
4. Cruciferous Vegetables
- Broccoli: High in vitamins C and K, fiber, and sulforaphane.
- Brussels Sprouts: Rich in vitamins C and K, fiber, and antioxidants like kaempferol.
- Cauliflower: Contains vitamins C and K, fiber, and various antioxidants.
5. Fruits
- Citrus Fruits: Oranges, lemons, and grapefruits are rich in vitamin C, flavonoids, and fiber.
- Pomegranates: Packed with vitamin C, potassium, and antioxidants like punicalagins.
- Grapes: Especially red and purple varieties, are rich in resveratrol and other polyphenols.
6. Other Vegetables
- Tomatoes: High in lycopene, vitamin C, and potassium.
- Bell Peppers: Especially red peppers, are rich in vitamins A and C, and beta-carotene.
- Sweet Potatoes: Contain beta-carotene, vitamins A and C, and fiber.
7. Herbs and Spices
- Turmeric: Contains curcumin, which has powerful anti-inflammatory and antioxidant properties.
- Ginger: Rich in gingerol, which has antioxidant and anti-inflammatory effects.
- Garlic: Contains allicin, which has antioxidant and antimicrobial properties.
- Cinnamon: High in cinnamaldehyde and other antioxidants.
8. Whole Grains
- Oats: Contain avenanthramides, which have anti-inflammatory and antioxidant effects.
- Quinoa: Rich in flavonoids, fiber, and various vitamins and minerals.
- Barley: Contains selenium, vitamins B and E, and various antioxidants.
9. Legumes
- Lentils: High in fiber, protein, and antioxidants like polyphenols.
- Black Beans: Contain anthocyanins, fiber, and protein.
- Chickpeas: Rich in fiber, protein, and antioxidants like quercetin.
10. Beverages
- Green Tea: High in catechins, especially epigallocatechin gallate (EGCG).
- Coffee: Contains various polyphenols and antioxidants.
- Red Wine: In moderation, provides resveratrol and other polyphenols.
Summary
Incorporating a variety of these nutrient-dense, antioxidant-rich foods into your diet can help protect against oxidative stress, support mitochondrial health, and improve overall cellular function. Eating a diverse range of these foods ensures you get a broad spectrum of antioxidants and other beneficial nutrients.
Test and Stop Guessing if you are FIT & HEALTHY from the inside out at a cellular level!
Measuring mitochondrial health can provide valuable insights into cellular function, energy production, and overall metabolic health. Functional lab testing for mitochondrial health typically involves a combination of direct and indirect assessments. Here are some of the key methods and tests used:
Assessment of Mitochondrial Function
A. VO2 Max Testing
- Purpose: Measures the maximum rate of oxygen consumption during intense exercise.
- Significance: Higher VO2 max indicates better mitochondrial function and aerobic capacity.
B. Lactate Threshold Testing
- Purpose: Determines the exercise intensity at which lactate begins to accumulate in the blood.
- Significance: A higher lactate threshold suggests better mitochondrial efficiency and capacity for sustained aerobic metabolism.
Laboratory Biomarkers
A. Blood Tests
- Lactate and Pyruvate Levels: Elevated levels may indicate mitochondrial dysfunction, as these metabolites accumulate when oxidative phosphorylation is impaired.
- Creatine Kinase (CK): High levels can suggest muscle damage or mitochondrial dysfunction.
- Coenzyme Q10 Levels: A vital component of the electron transport chain; low levels can indicate impaired mitochondrial function.
B. Urine Organic Acids Test (OAT)
- Purpose: Analyzes organic acids in urine, which are byproducts of metabolism.
- Significance: Abnormal levels of specific organic acids (e.g., succinate, fumarate, lactate) can indicate issues with mitochondrial pathways such as the Krebs cycle or oxidative phosphorylation.
C. Oxidative Stress Markers
- Malondialdehyde (MDA) and F2-isoprostanes: Indicators of lipid peroxidation and oxidative stress.
- 8-Hydroxy-2′-deoxyguanosine (8-OHdG): A marker of oxidative damage to DNA, including mitochondrial DNA.
Genetic and Molecular Testing
A. Mitochondrial DNA (mtDNA) Analysis
- Purpose: Identifies mutations, deletions, or copy number variations in mtDNA.
- Significance: Genetic defects can directly impact mitochondrial function and health.
B. Gene Expression Profiling
- Purpose: Measures the expression levels of genes involved in mitochondrial biogenesis and function, such as PGC-1α, NRF1, and TFAM.
- Significance: Altered expression of these genes can indicate changes in mitochondrial biogenesis and activity.
Functional Assays
A. High-Resolution Respirometry
- Purpose: Measures the oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) of isolated mitochondria or live cells.
- Significance: Provides detailed information on the functionality of the electron transport chain and overall mitochondrial respiration efficiency.
ATP Production Assay
- Purpose: Quantifies the amount of ATP produced by mitochondria under different conditions.
- Significance: Reduced ATP production can indicate compromised mitochondrial function.
Advanced Imaging Techniques
A. Electron Microscopy
- Purpose: Provides high-resolution images of mitochondrial structure.
- Significance: Structural abnormalities can correlate with functional impairments.
B. Fluorescent Imaging
- Purpose: Uses fluorescent dyes to assess mitochondrial membrane potential, ROS production, and mitochondrial mass.
- Significance: Changes in these parameters can indicate mitochondrial health or dysfunction.
Clinical Tests and Panels
Several commercial and clinical laboratories offer comprehensive panels specifically designed to assess mitochondrial health. These panels may include a combination of the above tests to provide a holistic view of mitochondrial function.
To measure mitochondrial health comprehensively, a combination of functional, biochemical, genetic, and imaging tests is often used. These tests can provide insights into mitochondrial efficiency, capacity, structural integrity, and the presence of oxidative stress. Regular assessment using these methods can help in early detection of mitochondrial dysfunction and guide interventions to improve mitochondrial health and overall cellular energy metabolism.
Mitochondria – What They Are, Why We Should Care, and How to Support Them Using Functional Medicine Strategies
by Dr.Laura DeCesarisMaster Clinical Nutrition, Chiropractic, A4M FellowshipThere’s a growing awareness that a long lifespan isn’t what most people truly want; rather, a long healthspan – meaning staying capable of living healthy, active lives while aging – is the penultimate goal. Currently, roughly 20% of the average person’s lifespan is spent in ill health due to chronic disease, and mitochondria may be important pieces of how to improve these statistics. Mitochondrial dysfunction has been linked to most chronic illnesses, including diabetes, accelerated aging, neurodegenerative disorders, obesity, and cancer.
What Are Mitochondria?
Mitochondria are parts of your cells that play an important role in generating energy to run metabolism from the carbohydrates and fatty acids you eat and store. These organelles contain their own DNA, which is inherited maternally. Mitochondria are surrounded by a double membrane with an intermembrane space and an interior compartment where the mitochondrial DNA is stored. Mitochondria also regulate important processes such as cell survival and calcium homeostasis, and they are often misregulated in chronic disease, making them an important target for longevity research.
The inner membrane of mitochondria houses an important set of proteins called the electron transport chain, where the breakdown products of carbohydrates and fatty acids are converted into ATP, the major source of energy for various metabolic reactions in the body. Through a process called oxidative phosphorylation, mitochondria generate ATP by utilizing the energy from the food we eat that is released via digestion. Your mitochondria create approximately your body weight in ATP each day!
Inherited disorders of mitochondria can cause severe metabolic problems leading to reduced ability to function or even death. However, these disorders are quite rare, and the more common relationship between mitochondria and overall health has to do with more subtle mitochondrial dysfunction that can play a role in chronic conditions such as insulin resistance, premature cellular aging, obesity, and Type II Diabetes.
Why Should We Care About Mitochondria?
Shifting the focus to mitochondrial health may be a game-changer when it comes to seeking optimal health and longevity. When mitochondria aren’t working well, the resulting loss of function can lead to excess fatigue and other symptoms that are common complaints in most chronic diseases. Since healthy mitochondria are needed to create ATP, any reductions in their function means less ATP available to provide energy for important metabolic reactions – leaving an individual feeling tired and not at their best.
Mitochondrial dysfunction has been linked to various chronic illnesses, including chronic fatigue syndrome and neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and Huntington’s disease. Since mitochondria play an important role in programmed cell death, or apoptosis, it’s theorized that unhealthy mitochondria can’t regulate cell death of errant cells as well in these conditions. Poor mitochondrial health has also been found to accelerate insulin resistance and the progression of metabolic syndrome due to the increased oxidative stress and inflammation that can build up when mitochondria are not performing well.
Mitochondria also play an important role in the aging process, contributing to processes such as cellular senescence, chronic inflammation, and the natural decline in stem cell activity that accompanies aging. Strategies that are aimed at improving mitochondrial quality, function, and biogenesis may have far-reaching effects on longevity and on healthspan if we can keep our mitochondria healthier for longer.
Functional Medicine Labs That Can Help Individualize Treatment for Mitochondrial Support
When it comes to supporting mitochondrial function, several laboratory tests can provide valuable insights into the health and efficiency of these essential cellular powerhouses. These tests can help identify potential imbalances, deficiencies, or dysfunction within the mitochondria. Here are some examples of laboratory tests that support mitochondrial function:
Organic Acid Test
Organic acid testing (OAT) helps assess nutritional deficiencies that can impact a person’s metabolic pathways, including energy production in the mitochondria. Organic acids are cellular byproducts produced through regular metabolic pathways that are required for survival, like energy production, protein and fat metabolism, and detoxification. The OAT has been shown in clinical studies to be an effective mitochondrial evaluation as part of an overall approach to mitochondrial disease.
CoQ10 Levels
CoQ10 is an important component of the electron transport chain within mitochondria. Inadequate CoQ10 levels can compromise the efficiency of energy production, leading to fatigue and other symptoms. Additionally, decreased CoQ10 levels can lead to increased oxidative stress in the cells since CoQ10 plays an important antioxidant role. Serum evaluation of CoQ10 can help determine if your levels are adequate.
Carnitine Levels
Carnitine is a compound that’s important for transporting fatty acids into the cell in order to be used for energy. Low carnitine may lead to issues with utilizing fats for fuel through oxidative phosphorylation in the mitochondria, showing up as symptoms like fatigue, hypoglycemia, steatosis, and even cardiomyopathy.
Oxidative Stress Markers
Mitochondrial DNA is a vulnerable target of oxidative stress when inflammation is high in the body. Once mitochondria are damaged, oxidative stress is further amplified as the damaged mtDNA can no longer effectively produce the proteins that compose the electron transport chain. If this process continues unchecked, mitochondria will undergo cell apoptosis and die, leading to symptoms of poor mitochondrial function such as metabolic dysfunction, fatigue, and others. Checking oxidative stress markers can help guide a functional medicine approach to supporting healthy mitochondria.
Fatty Acid Profile
A fatty acid profile is used to assess the balance between essential and non-essential fatty acids that are important for optimal metabolic health while also providing useful information on the ratio of omega-6 and omega-3 fatty acids, which can indicate overall inflammation. The mitochondrial membranes are made of these fatty acids, and composition can change with levels of various fatty acids. The function of mitochondria may be impacted by non-optimal levels of fatty acids, making it important to keep an eye on these biomarkers. For example, excessive omega-6 content may impair the function of the lipid membrane, affecting how glucose and fatty acids are transported to be used to make ATP within the mitochondria.
Supporting Mitochondria Through Functional Medicine Strategies
A functional medicine approach can be used to support healthy mitochondria.
Below are some common functional medicine strategies:
A nutrient-dense, anti-inflammatory nutritional approach is essential for optimal mitochondrial health and function. Creating ATP for energy is a metabolically demanding process that requires a variety of vitamins, minerals, and other nutrients to run efficiently. The Mediterranean-type diet is one such approach that has been found to limit oxidative stress and ameliorate damage to the mitochondria, making it a smart choice to support mitochondrial health.
Including foods and nutrients that help prevent oxidative damage are particularly important, such as antioxidants found in brightly colored fruits and vegetables and various herbs and spices, as well as foods rich in omega-3 fats such as coldwater fish and flaxseed.
Additionally, vitamins and minerals like vitamin C, zinc, magnesium, CoQ10, carnitine, creatine, and B vitamins are needed for optimal mitochondrial health.
Optimizing Mitochondrial Function With Supplements And Herbs
Below are some supplements that can help with mitochondrial optimization:
CoQ10
CoQ10 supplementation may be linked with alleviating inflammation and oxidative stress while decreasing symptoms of aging by impacting mitochondrial energy production.
Dose: 100mg 3 times per day
Duration: At least 8 weeks
Pyrroloquinoline Quinone (PQQ)
PQQ appears to impact mitochondrial biogenesis by increasing key regulators of the energy production pathways in mitochondria. It may be a helpful antioxidant to improve overall mitochondrial health and efficiency as part of an overall healthy lifestyle.
Dose: 20mg daily
Duration: 6 weeks
Omega-3 Fatty Acids
Omega-3 fish oil supplementation has been linked to improving the health of mitochondrial membranes while reducing reactive oxygen species characteristic of oxidative stress.
Dose: 2g EPA, 1g DHA
Duration: 12 Weeks
Alpha-Lipoic Acid
ALA is an antioxidant molecule that is involved in breaking down carbohydrates for energy in the mitochondria. Although, to date, there have been no controlled clinical trials done on ALA for mitochondrial disorders, the Mitochondrial Medicine Society (MMS) recommends ALA supplementation, and no safety issues have been documented.
Dose: up to 600mg/day
Duration: 6 months
Carnitine
Carnitine is involved in the transportation of long-chain fatty acids into the mitochondria to create ATP. It also supports the removal of toxic metabolites from the mitochondria.
Dose: less than 2g/day in short duration
Lifestyle Modifications For Mitochondrial Health
- One of the best lifestyle tools to improve mitochondrial health is regular physical activity and exercise.
- Exercise has been found to strongly induce PGC1-alpha, a compound that activates mitochondrial biogenesis and improves energy production. It should be noted that this is based on training intensity.
- While any form of movement is better than no movement, working in some higher-intensity exercise is going to make the biggest difference in getting the beneficial effects of improved mitochondrial biogenesis.
- Getting great sleep and sticking to a regular sleep routine is important for mitochondrial health as well since mitochondrial function is linked to the circadian rhythm (our sleep-wake cycle).
- A lack of sleep can negatively impact how well the mitochondria can work.
- Sleep is an important time for the body to “clear out” older, damaged mitochondria and turn over new mitochondria for optimal energy production.
- Managing stress levels is also important for mitochondrial health.
- When under stress, mitochondria can actually swell, leading to impairments in transport across the membranes and in the function of the electron transport chain – meaning they can’t produce energy as well.
- Mitochondria are responsible for meeting the large energy demands of a prolonged stress response, meaning that poorly managed stress may place such a demand on the mitochondria that it can become difficult for mitochondria to maintain homeostasis and effectively do their job.
Summary
- Mitochondria are small cellular organelles that play a massively important role in how your metabolism runs efficiently, how you produce energy, and how you meet the demands you place on your body day in and day out.
- A functional medicine approach to supporting mitochondria may include functional medicine labs to assess underlying contributors to poor mitochondrial function, nutritional interventions, supplementation, and lifestyle strategies like optimizing sleep and stress management.
- The healthier your mitochondria are, the more likely you are to experience optimal health and well-being.
- The information provided is not intended to be a substitute for professional medical advice.
- Always consult with your doctor or other qualified healthcare provider before taking any dietary supplement or making any changes to your diet or exercise routine.
- https://www.rupahealth.com/post/mitochondria-what-they-are-why-we-should-care-and-how-to-support-them-using-functional-medicine-strategies







